NOT THE RIGHT ROLE?
Open application
At ROTOP, we are always looking for people who work with commitment, curiosity, and a strong sense of responsibility.
Even if there is currently no suitable position advertised, we welcome your open application.
Show us who you are – we will carefully review your profile and get in touch as soon as a suitable opportunity arises.

WHY YOU SHOULD WORK FOR ROTOP
Jobs at ROTOP
ROTOP stands for innovative medicines and excellent solutions in radiopharmacy. Join a team where creativity, responsibility, and collaboration drive new approaches every day. Whether in research, production, administration, or sales, ROTOP offers exciting opportunities for you to apply your skills and grow professionally. Work on innovative projects, shape processes, and help actively drive the future of radiopharmacy.
Become part of a company that values quality, safety, and progress — and take an active role in shaping your career.
MEET OUR TEAM
We are ROTOP

Anne-Marie | Head of Manufacturing
I actually came across the job posting quite by chance – and now I’ve celebrated my 5-year anniversary with ROTOP! I was particularly interested in the radiation protection aspect, having already completed a related course during my studies. I was therefore glad that my application was welcomed here and that the interview process was very pleasant.
Martin | Regulatory Affairs Specialist
I trained as a Chemical-Technical Assistant (CTA). ROTOP offered me a role that combined lab work with regulatory tasks, which really caught my interest since I had no prior experience in drug approval—and there’s no formal training program for it. So at first, it was very much a case of “learning by doing,” which gave me a great opportunity to develop my skills.


Kristin | Accounting Manager
Before joining ROTOP, I worked at a tax office handling multiple clients at the same time. It was very hectic, and I wanted to move to a role where I could make a meaningful contribution within a single company. When we moved closer, I came across ROTOP. The application process was straightforward and quick, so nothing stood in the way of starting right away.
MEET OUR TEAM
We are ROTOP

Anne-Marie | Head of Manufacturing
I actually came across the job posting quite by chance – and now I’ve celebrated my 5-year anniversary with ROTOP! I was particularly interested in the radiation protection aspect, having already completed a related course during my studies. I was therefore glad that my application was welcomed here and that the interview process was very pleasant.

Martin | Regulatory Affairs Specialist
I trained as a Chemical-Technical Assistant (CTA). ROTOP offered me a role that combined lab work with regulatory tasks, which really caught my interest since I had no prior experience in drug approval—and there’s no formal training program for it. So at first, it was very much a case of “learning by doing,” which gave me a great opportunity to develop my skills.

Kristin | Accounting Manager
Before joining ROTOP, I worked at a tax office handling multiple clients at the same time. It was very hectic, and I wanted to move to a role where I could make a meaningful contribution within a single company. When we moved closer, I came across ROTOP. The application process was straightforward and quick, so nothing stood in the way of starting right away.
WHAT WE OFFER YOU
Your benefits at ROTOP

YOUR CAREER WITH US
ROTOP as Employer
Curious about what it’s like to work at ROTOP? Our employer brochure gives you a compact overview of our work environment, teams, and career opportunities – from apprenticeships and dual study programs to specialist and leadership careers.
Discover what makes ROTOP a great place to work and download the german brochure as a PDF now.
FROM LAB TO IMPACT!
Start Your Apprenticeship at ROTOP!
YOUR WAY TO ROTOP
Our recruitment process – simple, transparent, personal
At ROTOP, we value a clear and fair application process that is transparent and easy to follow for you.
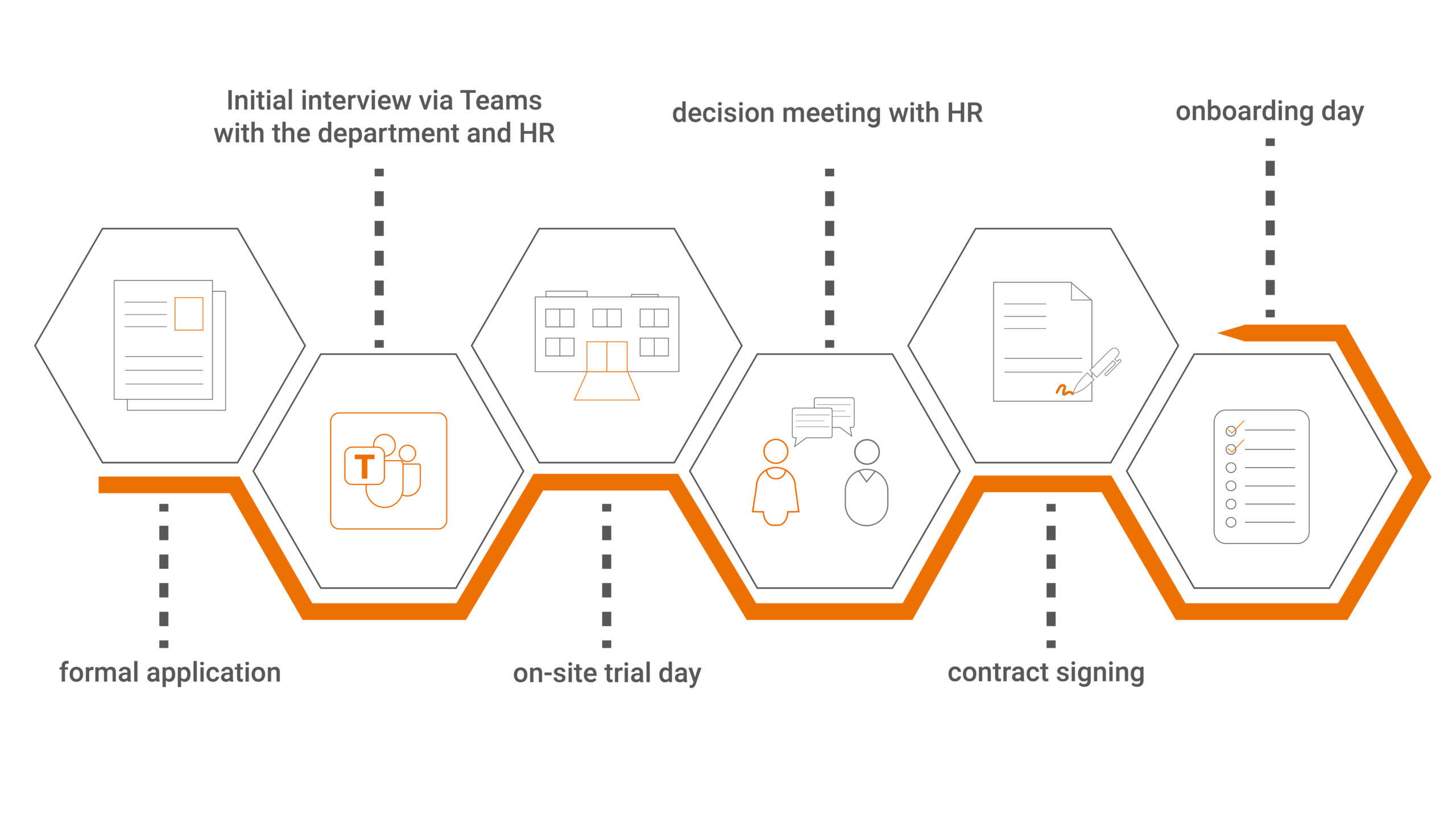
Our goal is for you to feel welcome from the very beginning, experience our company culture, and get off to a great start in your new role.
Questions about our company?
Contact us:
Sven Schäfer | Recruiting
+49 351 26310201
application@rotop-pharmaka.com
(Attachments in PDF format)
We value diversity and equal opportunities.
Applications without a photo are warmly welcome.

MEET US IN PERSON
Upcoming Career Events
YOUR WORKPLACE
Location: Dresden-Rossendorf
ROTOP is located on the campus of the Helmholtz-Zentrum Dresden-Rossendorf (HZDR), one of Germany’s leading research sites. The location combines a strong network of expertise with an inspiring working environment.
Dresden-Rossendorf is part of a vibrant innovation cluster where research institutes, start-ups, and companies from the fields of biotechnology, nuclear medicine, and radiopharmacy collaborate closely.
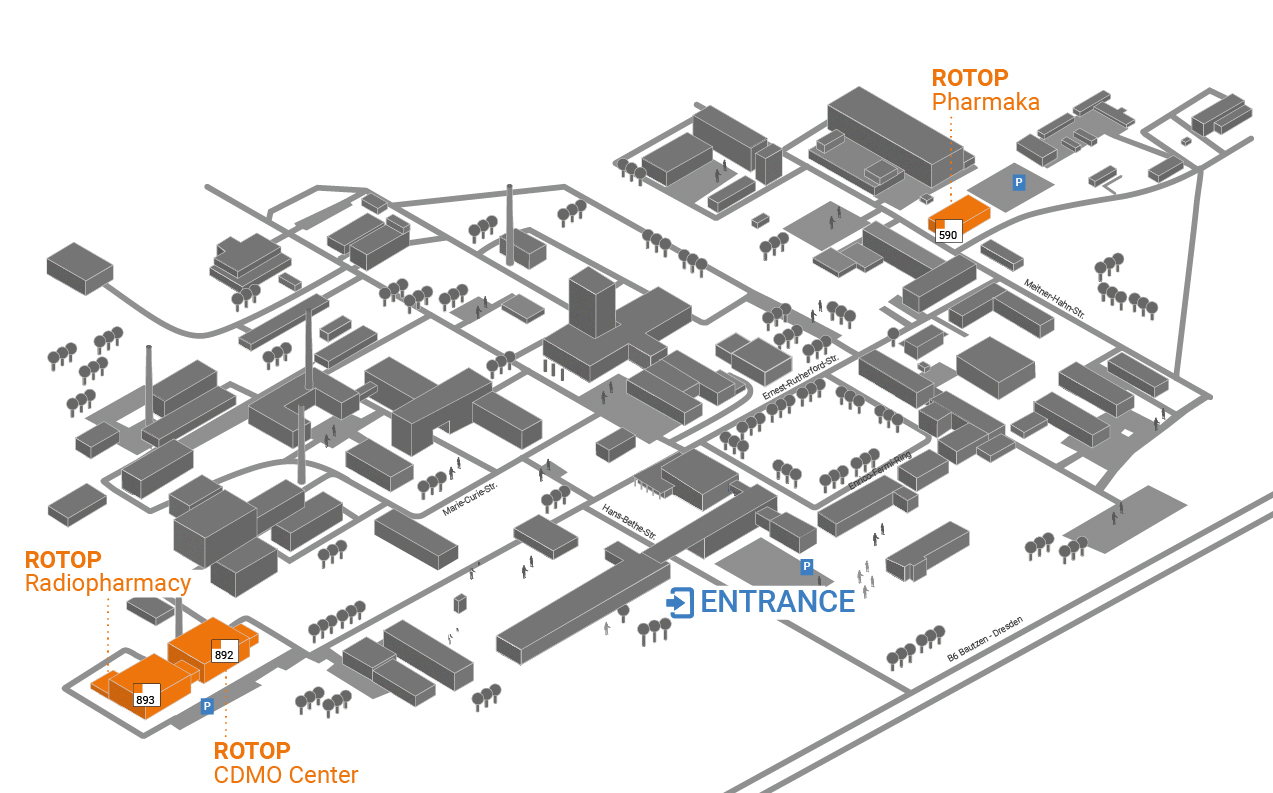

Short distances and the exchange within the nuklid® network create ideal conditions to quickly turn ideas into forward-looking technologies.
The location is easy to reach: the campus offers free parking and well-developed cycling paths. The surrounding area provides plenty of nature and opportunities for outdoor activities. At the same time, the city center of Dresden is easily accessible for culture, dining, and urban justify.
The HZDR campus is known as a “Green Campus” and has been consistently focusing on sustainability and resource conservation since 2002 — an approach that also benefits our site.
Here, employees work in a dynamic and international environment that encourages innovation, teamwork, and creativity — right at one of Germany’s most forward-looking science and technology locations.
FAQ
Frequently Asked Questions